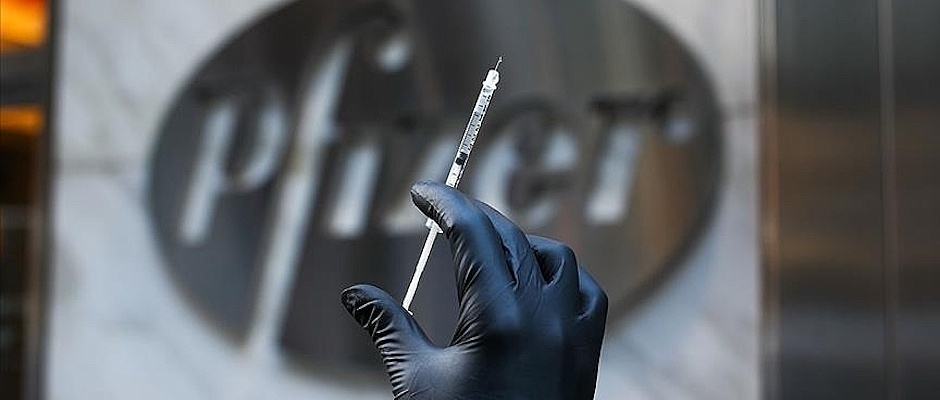
The UK's Medicines and Healthcare products Regulatory Agency announced on Wednesday that those with a history of "significant" allergic reaction to drugs, food or vaccines should not take the Pfizer-BioNTech coronavirus vaccine.
The decision came after two staff members of the National Health Service who were given the vaccine experienced an "anaphylactoid reaction," referring to allergic symptoms. Both had histories of allergies and are now reportedly recovering.
The UK became the first country in the world to deploy the coronavirus vaccine after the Pfizer-BioNTech jab was approved by the MHRA last week.
Dr. June Raine, the head of the MHRA, told a parliamentary hearing: "We know from the extensive clinical trials that this was not a feature, but if we need to strengthen our advice now that we have had this experience in vulnerable populations, the groups selected as a priority, we get that advice out immediately."
A spokeswoman for Pfizer said they had been advised by the MHRA of two reports "that may be associated with allergic reaction" due to being administered their vaccine.
"As a precautionary measure, the MHRA has issued temporary guidance to the NHS while it conducts an investigation in order to fully understand each case and its causes. Pfizer and BioNTech are supporting the MHRA in the investigation," she added.
"In the pivotal phase three clinical trial, this vaccine was generally well tolerated with no serious safety concerns reported by the independent Data Monitoring Committee. The trial has enrolled over 44,000 participants to date, over 42,000 of whom have received a second vaccination."


 CTCA UK Condemns the Political Forcing Out of Afzal Khan MP for Engaging with Turkish Cypriots
CTCA UK Condemns the Political Forcing Out of Afzal Khan MP for Engaging with Turkish Cypriots Tatar: “Reaction to MP’s TRNC visit is yet another stark example of the Greek Cypriot leadership’s primitive and domineering mentality”
Tatar: “Reaction to MP’s TRNC visit is yet another stark example of the Greek Cypriot leadership’s primitive and domineering mentality” Margaret Greer has been sworn in as the new Mayor of Enfield
Margaret Greer has been sworn in as the new Mayor of Enfield Prime Minister Keir Starmer's 2025 Easter message
Prime Minister Keir Starmer's 2025 Easter message Team Enfield ranks fifteenth the in London Youth Games
Team Enfield ranks fifteenth the in London Youth Games Parking enforcement boosted with more officers on patrol in Enfield
Parking enforcement boosted with more officers on patrol in Enfield Ersin Tatar meets with President Erdoğan
Ersin Tatar meets with President Erdoğan President Ersin Tatar Holds Ministerial-Level Meeting with UK Minister for Europe
President Ersin Tatar Holds Ministerial-Level Meeting with UK Minister for Europe UEFA Europa League and UEFA Conference League draws to be combined into one single show
UEFA Europa League and UEFA Conference League draws to be combined into one single show EuroLeague schedule for 2025-26 season announced
EuroLeague schedule for 2025-26 season announced Zeynep Sonmez becomes first Turkish tennis player to reach third round at Wimbledon
Zeynep Sonmez becomes first Turkish tennis player to reach third round at Wimbledon European champions Arsenal Women will play all of their league matches at the Emirates Stadium
European champions Arsenal Women will play all of their league matches at the Emirates Stadium Enfield Labour welcomes the completion of A10 average speed cameras extension.
Enfield Labour welcomes the completion of A10 average speed cameras extension. TfL opens 2025 grants for community groups to encourage more walking, cycling and active travel in the capital
TfL opens 2025 grants for community groups to encourage more walking, cycling and active travel in the capital Fethiye Launches International Digital Tourism Campaign with UK-Based Publisher
Fethiye Launches International Digital Tourism Campaign with UK-Based Publisher Highlights from the 3rd Trans-Caspian Connectivity Conference in London
Highlights from the 3rd Trans-Caspian Connectivity Conference in London